"Novel Hybrid & Inorganic Perovskites for Solar cells"
In order to convert the enormous amount of solar energy into usable electric energy,
solar cells are utilized which work under the principle of photovoltaic effect.
However the efficiency of such solar cells decreases as the ambient temperature
increases due to the inherent indirect-band gap nature. Hence alternative materials
are researched to circumvent these problems.
Metal halide organic-inorganic perovskites have many favorable properties that are applicable
for next generation solar cells. For example MAPbI3 is the champion among hybrid perovskites
which is made using the low-temperature spin coating technique.
Hybrid perovskite is used as absorber material in the solar cells. The hybrid perovskites has
the structural constituents comprising of both organic and inorganic components. General
chemical
formula of the compounds is ABX3. The A-site is occupied by organic cation such as
Methyl-ammonium
(MA) and Formamidinium (FA) or inorganic cation such as Cesium (Cs); B-site by divalent group
IV-a
cation such as Pb, Sn and the X-site is usually occupied by halide anion such as Cl, Br and
I.
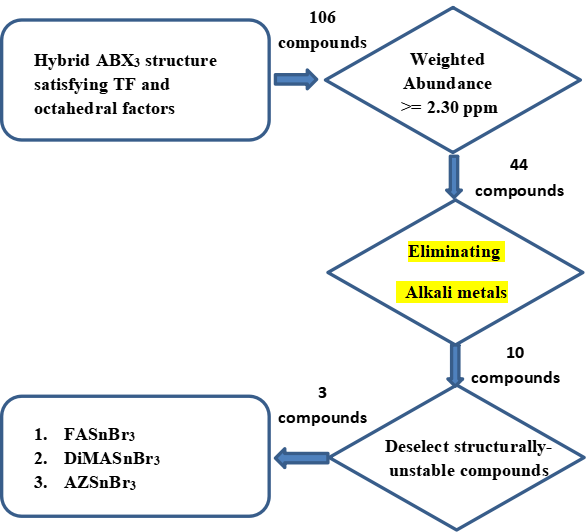
We have used a novel approach to search for new perovskite materials based on permutation of the
perovskite constituents suitably chosen from the periodic table. The permutations were started
with 18 organic molecular cations, 9 divalent metal ions and three halides. Considering the
stability factors, 106 compounds were picked out of 486 structures matching the tolerance factor
and octahedral factors which resulted in good prediction for preliminary screening before high
computational DFT calculation.
We predicted that the broad bandgap tuning in Cs2SnGeX6 (X = I, Br, Cl) with the closer lattice
parameters can enable the fabrication of functionally graded material with varying halide
constituents
leading to tandem solar cells that can absorb a broad range of the solar spectrum. Also, the
hybridization
interaction between Sn-s and X-p orbitals, leading to a highly dispersed band (~2.5eV) at the
valence
band maximum, giving rise to holes with light effective mass.
Quick Links
Reach Us
Sir CV Raman Block , Anna University.
C-MaIn@annauniv.edu
044-22359938
© C-mAIn. All Rights Reserved. Designed by HTML Codex. Maintained by Scholars of C-mAIn.